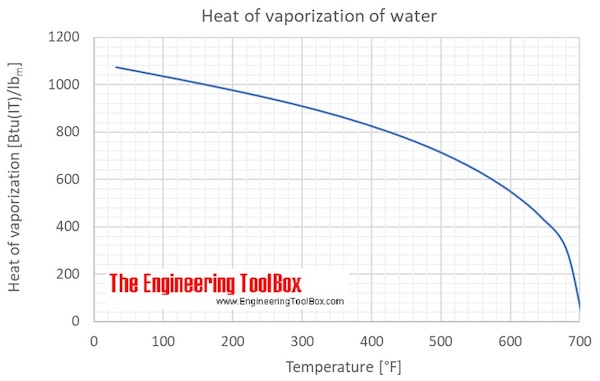
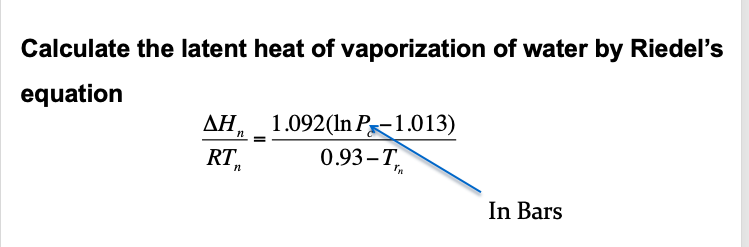
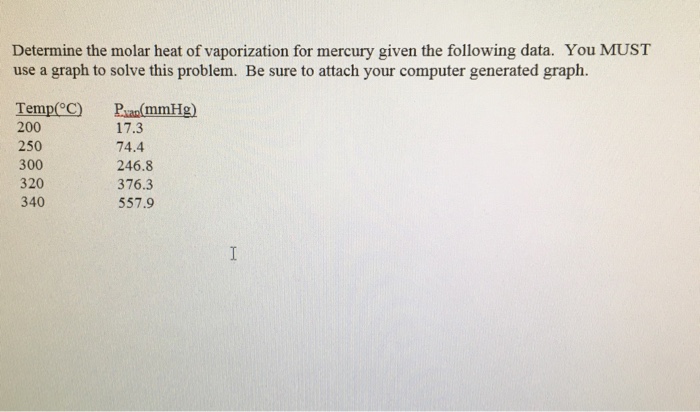
![SOLVED: Using the Clausius-Clapeyron equation O iie data provided in %he problem to graphically determine (on excc] spread shect) the heat of vaporization Of nitrogen. Graph the data below manner used detenmnine SOLVED: Using the Clausius-Clapeyron equation O iie data provided in %he problem to graphically determine (on excc] spread shect) the heat of vaporization Of nitrogen. Graph the data below manner used detenmnine](https://cdn.numerade.com/ask_images/be895267f44e437c8bb031c3001bb014.jpg)
SOLVED: Using the Clausius-Clapeyron equation O iie data provided in %he problem to graphically determine (on excc] spread shect) the heat of vaporization Of nitrogen. Graph the data below manner used detenmnine

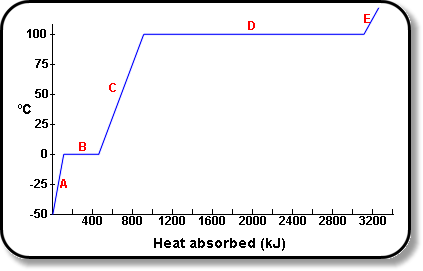
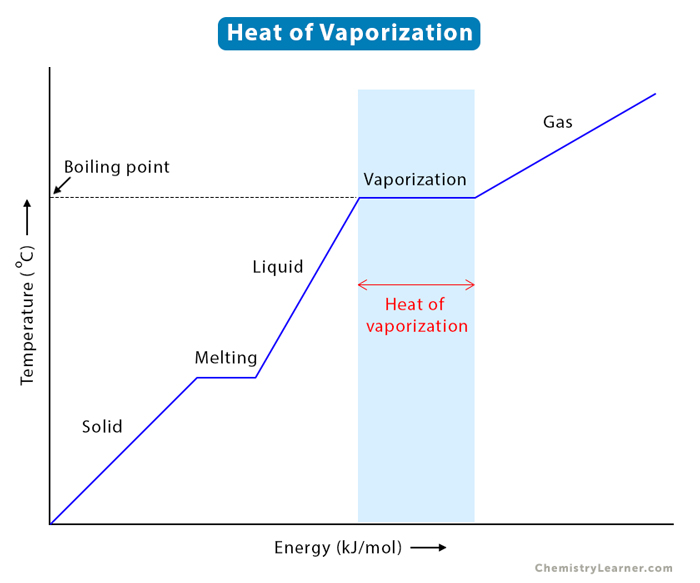
Using Heat of Fusion or Vaporization to Find the Heat Needed to Melt or Boil a Substance | Chemistry | Study.com